The project live streams AWS public datasets/XETRA dataset using Apache Spark along with WebSocket. starts Big Data Series 1: Data Live with Spark. The project headlines globe events collected by GDELT project. Linux virtual boxes built from scratch using instructions from Linux from Scratch 8.4 and Beyond Linux from Scratch 8.4 are available to Features of Stoichiometry Plus 2.0 can be found here The new release adds Solution Preparation to the calculator collections. Between decimals and fractions, lossless fractions should be the first choice. 5) Integers, decimals and fractions can all be used in equations. 4) Use * to formulate composite compounds, for example: CuSO4*3H2O, CuSO4*7/2H2O, CuSO4*0.5H2O. The following conventions are used for thermodynamics calculations:ġ) Enclose charges and states in order in curly brackets. Require complete information, such as state, charge, phase etc., for each chemical. The equilibrium vapor pressure P of liquid water at a temperature T can be accurately estimated using the Tetens equation: P 0.61078 exp ( 17.27 T T + 237. Thermodynamics calculations (Reaction Thermodynamics and Equilibrium Composition) Both CuSO4*1/2H2O and CuSO4*0.5H2O will produce the same results.ī) Conventions for Thermodynamics Calculations 3) Both fractions and decimals are acceptable. To make the multiple (2 in this case) just for CuSO4, you can write the formula as (CuSO4)2*0.5H2O. 2) A number at the beginning of a formula is interpreted as multiple of the whole formula, eg: 2CuSO4*0.5H2O = (CuSO4)2*H2O. 
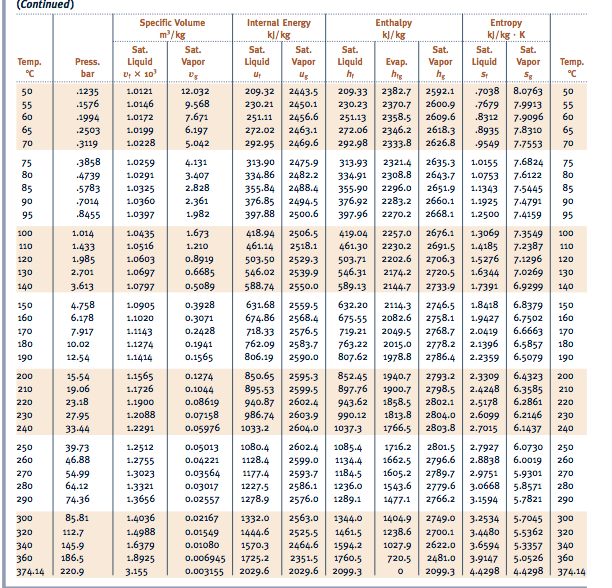
The below tables and graphs is computed at 1atm 1.01325Bar (A) Temperature. This site also contains a calculator capable of calculating the properties as function of temperature and pressure. However, when a formula is a composite compound, followingġ) Using * to formulate composite compounds such as CuSO4*0.5H2O. The thermodynamic property table and the graphs for water below is based on the industrial formulation by IAPWS-IF97. In general, you just need to type in a formula and then go. In this review, we provide a comprehensive summary of published uses of WaterMap, including applications to lead optimization, virtual screening, selectivity analysis, ligand pose prediction, and druggability assessment.ĭruggability Kinetics Lead optimization Solvation Specificity Thermodynamics Water WaterMap.A) Conventions for Formula Weight Calculation and Equation Balancer You can convert that easily into a mass now. From the reaction equation you can now determine the amount of substance of water, that will be produced. This method can be used to assess the solvent contributions to ligand binding affinity and to guide lead optimization. You have been given the temperature T 25 C 298.15 K and pressure p 100 k P a and the volumes for the reactants, so you can calculate the amount of substance for propane and oxygen. WaterMap is a molecular dynamics-based computational method that uses statistical mechanics to describe the thermodynamic properties (entropy, enthalpy, and free energy) of water molecules at the surface of proteins. You will not need to be able to calculate the enthalpy directly in chemistry, we are only interested in the. Furthermore, understanding the energetic effects of solvation and desolvation on binding requires an analysis of the thermodynamic properties of solvent involved in the interaction between ligands and proteins. The value of C for water is 1.00 cal/g-C. While crystallography, NMR, and other experimental techniques can assist in determining the structure of water networks in proteins and protein-ligand complexes, most water molecules are not fully resolved and accurately placed. 
The ability to accurately characterize the solvation properties (water locations and thermodynamics) of biomolecules is of great importance to drug discovery.
0 Comments
Leave a Reply. |
 RSS Feed
RSS Feed
